Publications
Publication Date:
23/04/2024
Author:
Musa Odongo
Richard Kajubi
Anthony Nuwa
Craig Bonnington
Kevin Baker
Maureen Nakirunda
Tonny Kyagulanyi
Jane Nabakooza
David Salandini
Jimmy Opigo
Godfrey Magumba
Jane Achan
Damian Rutazaana
Denis Rubahika
James Tibenderana
Type:
Poster

Adverse events and reporting mechanisms for seasonal malaria chemoprevention: Insights from a survey in Karamoja region, Uganda
Publication Date:
23/04/2024
Author:
Musa Odongo
Richard Kajubi
Anthony Nuwa
Craig Bonnington
Kevin Baker
Maureen Nakirunda
Tonny Kyagulanyi
Jane Nabakooza
David Salandini
Jimmy Opigo
Godfrey Magumba
Jane Achan
Damian Rutazaana
Denis Rubahika
James Tibenderana
Type:
Poster

Publication Date:
23/04/2024
Author:
Musa Odongo
Richard Kajubi
Anthony Nuwa
Craig Bonnington
Kevin Baker
Maureen Nakirunda
Tonny Kyagulanyi
Jane Nabakooza
David Salandini
Jimmy Opigo
Godfrey Magumba
Jane Achan
Damian Rutazaana
Denis Rubahika
James Tibenderana
Type:
Poster
Publication

Adverse events and reporting mechanisms for seasonal malaria chemoprevention: Insights from a survey in Karamoja region, Uganda
Publication Date:23/04/2024
Author:
Musa Odongo
Richard Kajubi
Anthony Nuwa
Craig Bonnington
Kevin Baker
Maureen Nakirunda
Tonny Kyagulanyi
Jane Nabakooza
David Salandini
Jimmy Opigo
Godfrey Magumba
Jane Achan
Damian Rutazaana
Denis Rubahika
James Tibenderana
Type:
Poster
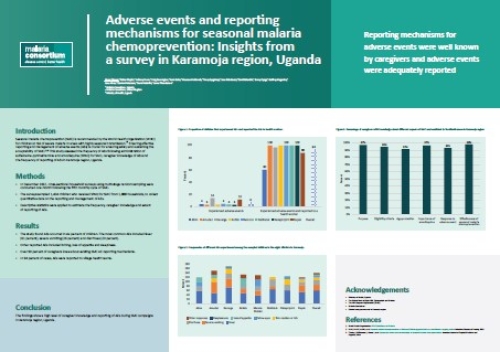
Reporting mechanisms for adverse events were well known by caregivers and adverse events were adequately reported.
Seasonal malaria chemoprevention (SMC) is recommended by the World Health Organization (WHO) for children at risk of severe malaria in areas with highly seasonal transmission. Ensuring effective reporting and management of adverse events (AEs) is crucial for ensuring safety and sustaining the acceptability of SMC. This study assessed the frequency of AEs following administration of sulfadoxine-pyrimethamine and amodiaquine (SPAQ) for SMC, caregiver knowledge of AEs and the frequency of reporting of AEs in Karamoja region, Uganda.
This poster was presented at the Multilateral Initiative on Malaria (MIM) Society's 8th Pan-African Malaria Conference.
Country: Uganda
Keywords: Malaria | SMC | Treatment
« Back to Publications